The examination of semen consists of smell, feel and fluorescence. When seminal stain is fresh or dried, its smell is characteristic in nature. When it dries, it has a rough feeling like a dried starch solution and has an uneven surface and also the contours are characteristics. The fluorescence of semen stains is also characteristic and it is affected by handling, quantity and age of the stain.
Physical Examination
During the physical examination of seminal fluid, an alternative light source may help to detection of some findings like injuries which may be invisible to the naked eye
Visual Examination
The suspected area or articles related to crime is examined visually. Semen is a thick, yellowish white secretion which is glairy, opalescent in nature, it has a characteristic odor which is commonly known as seminal odor. Its texture is starchy, when touched.
UV/IR Examination
When the suspected area is examined under ultraviolet rays in dark, it gives bluish white fluorescence and it occurs due to presence of choline in semen. The fluorescence of seminal flaid depends on its freshness and quantity,
Chemical examination of semen
Acid Phosphatase Test
Acid Phosphatase test is one of the best and generally used tests for presumptive identification of semen. Lundquist (1945), in Copenhagen, reported that a reasonably large amount of prostatic acid phosphatase present in human semen used as the identification of semen in medico-legal condition. On the basis of acid phosphatase enzyme present in semen is much higher concentration than other body fluids if the test is positive, it suggests that the semen is present. Its sensitivity is 400 times higher in semen than other body fluids. A suspect who is involved in sexual offence, either azoospermic or vasectomized is easily identified method a seminal stains, the stability of acid phosphatase is affected by temperature.
Reagents
Sodium Chloride-250 ml
Glacial Acetic Acid-5 ml
Anhydrous Sodium Acetate-12 gm
Brentamine Fast Blue-B salt, 0.5% (w/v)-250 gm
Calcium Alpha-naphthyl Phosphate 0.25% (w/v) – 125 gm
1% Teepol solution – 10 ml
Distilled water – 900 ml
Buffer Solution
A buffer solution is prepared by mixing Sodium Chloride, Glacial Acetic Acid, Sodium Acetate and distilled Water.
Reagent – 1
Mixture of Buffer solution 50 ml and Calcium Alpha-Naphthyl Phosphate. 0.25% (w/v)-125
Reagent 2
Mixture of buffer solution-50ml and Brentamine fast blue-B salt, 0.5% (w/v)-250 gm.
Procedure
Take a filter paper and transfer the staining material on it. Add 1-2 drops of reagent-1 and give it to rest for 30 seconds, after that again add 1 drop of meagent-2. After 20 seconds the appearance of filter paper is purple in color on the corresponding area which indicates that the stain is semen is observed.
Mechanism of Acid Phosphatase Test
Enzyme that present in stain is acid phosphatase and react with Calcium Alpha-Naphthyl phosphate which breaks into calcium and Alpha-Naphthyl Phosphate and after that Brentamine fast blue-B react with Alpha-Naphthyl phosphate gives Purple colour.
Creatine Phosphokinase
Griffiths and Lehman (1964), reported that the medico-legal identification of seminal stains is based on the presence of high levels of creatine phosphokinase in the semen. The range of creatine phosphokinase is from 385-14000 units ml which is much higher than other biological fluids examined and also including normal and pathological serum faeces, gastric juices, urine and veginal secretion etc. The reaction is measured by use of ADP and creatine phosphate as an enzyme. In 1968, Suyama et al., discovered that seminal acid phosphatase hydrolyzes creatine phosphate in the absence of ADP. At this stage when adding a specific SAP inhibitor, the activity is comparatively low of creatine phosphokinase present in semen.
Mechanism of Creatine Phosphokinase
The changing of creatine and adenosine miphosphate (ATP) into phosphocreatine and adenosine diphosphate (ADP), is catalysed by creatine phosphokinase
Observation
As the age of stains is increasing day by day, the activity of creatine phosphokinase is declined like 24 hours of age after ejaculation, the mean activity of the stain is 142 units per md, after one month of age of stains the mean activity has declined to 135 and after six months of age of stains the mean activity is declined to 80.
Spermin Crystal Test
Spermine crystal was first introduced by Anton Van Leevenbook (1677), and the chemical test for detection of crystals of spermine from the semen was given by Barberio (1905). This test is based on the formation of spermine crystal due to the chemical reaction between spremine present in semen and picric acid which is added as a reagent.
Reagent
Saturated solution of picric acid in distilled water or ethyl alcohol.
Procedure
Take a small amount of semen sample and allow it to treat with the reagent of Examined under the microscope.
Observation
Formation of characteristic crystal of Spermine pierate indicates the presence of semen. Crystals are yellow colorod, needle shaped or sometimes hollow boat shaped, Generally 10-30 microns in their size and overlapped each other.

Choline Crystal Test
This test was first introduced by Dr. Florence (1886), for identification of Choline crystal from the semen sample. This test is based on the formation of crystals of choline iodide because of the chemical reaction between choline present semen with iodine in reagent.
Reagent
Potassium Jodide-1.65 gm
Iodine-2.54 gm
Distilled water _ 30 ml
Procedure
A small portion of stain is soaked in warm distilled water and acidified with dilute Ir acid. Take 1-2 drops of extract on a microscopic slide, further added a drop of Florence reagent.
Now examined under the microscope
Observation
Formation of crystals of choline per iodide with characteristic features. Crystals are dark brown coloured, rhombic or needle shaped and are large and small in their size such as large crystals having length 80-120 microns while small crystals having length 20-35 microns. Crystals are isolated from each other.
Detection of Choline and Spermine through Thin Layer Chromatography
Yano (1970), described the method for detection of presence of choline and spennine in seminal stains. This method is fairly sensitive, rapid and gives positive test of seminal stains of about five years old.
Materials Required
TLC plate. Developing chamber, Potassium lodoplatinate reagent. Dragendroff magent, Micropipette etc
Solvent System
I N Hydrochloric Acid.
Dragendroff Reagent
Stock Solution
50% Solution of Potassium lodide in 100 ml Water
Bismuth Sub-nitrate-1.5gm
Glacial Acetic Acid-20 ml
Distilled Water-80 ml
Working Solution
10 ml of stock solution 15 ml of glacial acetic acid add water to make it 100 ml.
Stock Solution (Potassium Indeplatinate!
10% Potassium Chloride-5 ml
4% Potassium lodide -70 md
Working Solution
Take stock solution and make it up to 100 ml with distilled water.
Procedure
With the help of micropipette, a drop of test sample and controlled sample is spotted at defined distance on the TLC plate. The plate is now put inside the developing chamber in which the solvent system is already there and left it for 20 minutes. After that TLC plate is taken out and dried with air dry, then cooled. Now the lower part of the plate is sprayed with “Dragendroff reagent”, for the detection of “Choline Spot” and the upper is part of the plate is sprayed with “Potassium lodoplatinate reagent, for the detection of “Spermine Spot”.
Observation
When spraying “Dragendroff reagent” on the TLC plate, the appearance of yellow colour is indicating the presence of “Choline” and when spraying “Potassium lodoplatinate reagent”, the appearance of brown colour is indicating the presence of “Spermine” The “Retardation Factor” of Choline and Spermine when calcolated, gives the values are 0.4 and 0.7 respectively.
Prostate Specific Antigen (P30-Test)
Sensabaugh (1977-78), detected the test of the isolation of P30 protein from the seminar FANOS. This protein is antigenic in nature and at the time of collecting the suspected semen sample from the veginal washing, it is not always detectable because of its high dilution rate. This test is also a very useful presumptive test of serminal fluids to find out the human prostate-specific, P30 protein.
Procedure
Take a small cut piece of stained sample which is incubating in distilled water or normal saline solution and place it for 2-3 hours or overnight at a temperature of 4 degrees for rehydration. Now conduct the process of centrifugation for 3 minutes at maximum angular velocity, further separating the centrifuged liquid, so the liquid can retain as it is and this extracted liquid used for the testing of P30 which is present in stain. The remaining precipitate can be used for slide preparation or DNA extraction for further analysis
Mechanism of P30-Test
The mechanism behind this test is, it works on Antigen-Antibody reaction and the action taking place on the test site of the P30 card with the antibody which is present on the P30 card and the antigen present in the sample. The formation of a chain when antigen and antibody bonded to each other, causes a pink coloured dye band which is visible on both the positive and negative result.
Observation
The results shown in two ways
- When the pink dye band is shown in the control region as well as in the test region, the result is showing positive appearance of P30 in seminal fluid.
- When the pink dye hand is shown only in the control region, it shows a negative result of P30 in seminal fluid.
Microscopic Examination for Spermatozoa
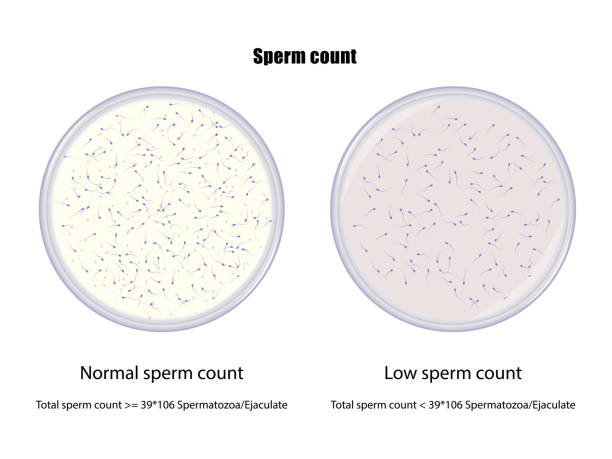
The microscopic examination of seminal stains is one of the confirmatory tests which clearly identify that the suspected stain is semen or any other fluid on the basis of its characteristic features and the relative quantity of spermatozoa and epithelial cells can also be analyzed. The sperm count in semen are of different types and can be seen through microscopic examination.
Oligospermia-Spermatozoa is countless in this category.
Azoospermia-Spermatozoa are little in amount and in deformed shape in this category.
Aspermia- Spermatozoa are absent in this category,
LDH Iso-Enzyme
The advantage of presence of LDH isoenzyme in stains is, it is stable in tropical conditions for over four weeks. The activity of LDH in human seminal fluid was first introduced by Macleod and Wroblewski (1958), and according to them the activity of LDH is lower than the whole seminal fluid, Blanco and Zinkham (1963), observed that every human semen has a unique LDH Isoenzyme.
The man who has low sperm count or sometimes has necrospermia condition and alteration is morphology of spermatozoa, the isoenzyme can also be detected. The fraction of activity of LDH and total seminal LDH is vary from one another and if a person has sperm count 22 x 10 per ml. The best method of examination of seminal LDH lsoenzyme, is Polyacrylamide Gel Electrophoresis and this might better for identification of spermatozoa in older semen stains.
Polyacrylamide Gel Electrophoresis (PAGE)
In this electrophoresis method we can separate the LDH isoenzyme because of composition of different amino acids
Materials Used
Pre-cast 7.5% polyacrylamide gel slab
Buffer tank (24 gm Tris base and 12 gm glycine/liter)
1 M Tris HCL pH-8.0
Tetrazolium-blue, I mg/ml
Phenazine Methosulphate, 1.5 mg/ml
IM Sodium Lactate
Procedure
Take a stained sample and wash out with the buffer tank and place the pre-cast gel into the electrophoretic chamber. Now load the sample into wells of the gel. The buffer tank runs into the reservoirs and connects with the electric cables, keeping the positive pole at the bottom. area. New start the power source at the current of 12 mA and run the electrophoresis at least for 90 minutes, after that switch off the power supply.
Separate the plates by inquire with the help of spatula and put the gel into the chamber which contains a developer solution and incubated at a temperature of 40 degrees, after 20 minutes the reaction takes place to the change in colour. In this reaction phenazine methosulphate acts as a primary election acceptor whereas tetrazolium blue acts as final electron acceptor and finally the gel is washed with water.
АР Іso-enzyme
Acid Phosphatase is one of the most important iso-enzymes which gives valuable information and identification of seminal fluid present at the scene of crime. The undertaking procedure of is-enzyme typing is known as electrophoresis, the process of separating different is-enzymes with the help of electric fields because their net charge is different.
Procedure
This process is carried out on different types of gel such as starch, agarose, polyacrylamide etc. These gels applied on the slides and placed into the chamber. The samples are now spread on the gel slide and powered on the electric current to the chamber throughout the gel. The proteins of negative charged moves on positive charged poles and the proteins of positive charged moves on negative charged poles. Same as proteins, it isoenzymes get charged, they also separate in the same way and once the process of electrophoresis has been done, the isoenzymes can be detected easily.
Staining Method
Generally the staining method is conducted for the purpose of to identify motility, quality etc. of the semen sample. The initial procedure of slide preparation is discussed here.
Procedure
Take a small cutting part from the stained area of the fabric which is moistened with 0.01 N Hydrochloric acid and kept for at least half-an-hour. Take out the individual threads of the fabric apart with the head of the stainless steel needle and squish out 1-2 drops of the liquid on the microscopic slide, preparing a thin smear on it. The slide is then allowed to air dry and then stained with any other of the following staining techniques and examined for the presence of the spermatozoa under microscope. The staining techniques are shown below:
Haematoxylin – Eosin
Haematoxylin Stain
Haematin-0.5 gm
Potassium Alum-0.5gm
Glycerine-30 ml
Distilled Water-70 ml
Eosin stain
Eosin-1 gm.
Distilled Water-100 ml
Observation
The posterior part of the head of stains is deep purple and the anterior part of the head of stains is pink in color.
Advanced Techniques-Cross over Electrophoresis
This is the traditional method for detection of P30 protein in semen and it is based on the identify the suspected stain antigen-antibody compounds. It plays a major role to is semen and widely used in most of the laboratories these days, further it takes more time to process and the sensitivity of this test is low.
Preparation of Plate
Take some microscopic slides further washed and cleaned with distilled water and detergent, after that make it dry. Numbering each and every slide on the right hand corner of the upper side consecutively.
Pre-coated plate
The glass slides were adhering with 1% of Agarose (o make sure that all the slides were pre coated with 0.2% Agaroses. Add 10 ml distilled water with 0.2% Agarose and boil upto dissolving, further applying this solution on the numbered glass slides and put it all to air dry. We can store these slides in the refrigerator and when required then frequently used.
Gel-coated Plate
The pre-coated slides are now put in the beaker and placed two tubes filled with 3.5 ml of 1% Agarose gel. Now put the beaker on a water bath, after that this liquefied solution is applied on each slide. Prevention takes place to overspill the liquid on the edges because of the pre-coating and surface tension property of the slides. Finally a flamen texture is formed on allowed to cool and toughen.
The Christmas tree Stain
The best confirmation test for the presence of semen is the positive visual identification of sperm cells (or spermatozoa) using the Christmas tree stain. Two main reagents are used consecutively to produce this distinctive stain: Picroindigocarmine stains the neck and tail portions of the sperm in green and blue, while Nuclear Fast Red (also known as Kernechtrot) gives the sperm heads a red color and the tips of the heads, an area known as acrosomal cap, a pink color Although this color pattern seems quite unique and may render sperm cells easily distinguishable under a microscope, sperm cells send to deteriorate quickly after ejaculation.
For more updates, subscribe to our blog